Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite.[6]
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, so fluorite is useful in making apochromatic lenses, and particularly valuable in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density.

Fluorspar Knowledge: From Fluorite to Modern Fluorine Chemistry
Fluorite (Fluorspar): The Foundation of Fluorine Chemistry
Fluorite (CaF₂) is a naturally occurring halide mineral that contains approximately 49% fluorine by weight. Its purity determines its market segment:
- Metallurgical grade (60–85% CaF₂): Used in steelmaking as a flux.
- Ceramic grade (85–95% CaF₂): Used in glass, glazes, and enamel.
- Acid grade (97%+ CaF₂): The most valuable grade, essential for producing fluorine-based chemicals.
Acid-grade fluorspar is processed into hydrofluoric acid (HF)—the chemical gateway to nearly all commercial fluorine compounds.
From Fluorspar to Hydrofluoric Acid (Fluorine Acid / Fluoric Acid)
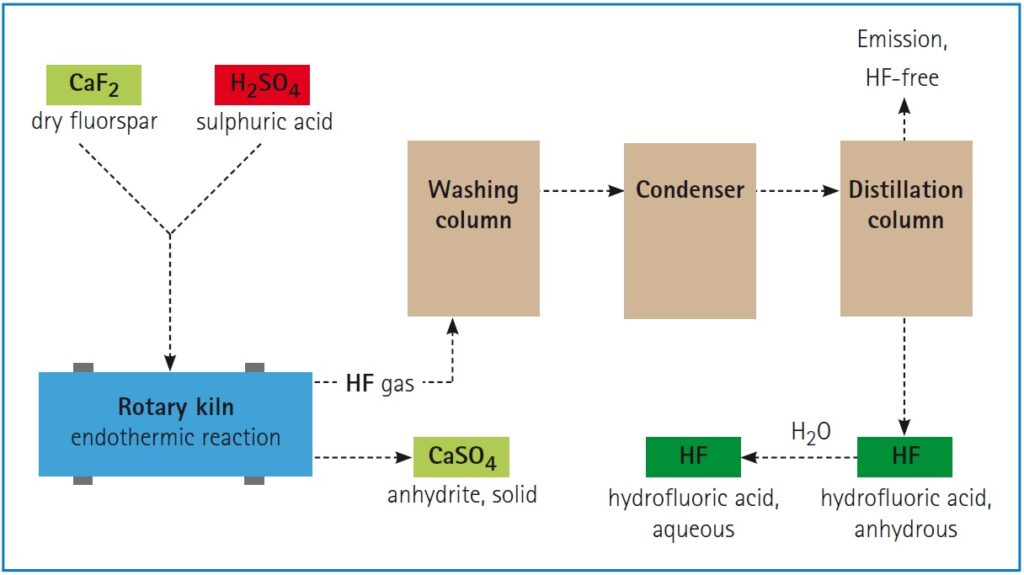

Fluorspar reacts with concentrated sulfuric acid to form hydrofluoric acid (HF), commonly referred to as:
- Fluorine acid
- Fluoric acid
- Hydrogen fluoride solution
Reaction:
CaF₂ + H₂SO₄ → 2 HF + CaSO₄
HF is a powerful, highly reactive acid used to create advanced fluorine-based chemicals such as fluorocarbons, fluoropolymers, and inorganic fluorides.
From Fluorspar to Hydrofluoric Acid (Fluorine Acid / Fluoric Acid)
Fluorspar reacts with concentrated sulfuric acid to form hydrofluoric acid (HF), commonly referred to as:
- Fluorine acid
- Fluoric acid
- Hydrogen fluoride solution
Reaction:
CaF₂ + H₂SO₄ → 2 HF + CaSO₄
HF is a powerful, highly reactive acid used to create advanced fluorine-based chemicals such as fluorocarbons, fluoropolymers, and inorganic fluorides.
Fluorine: The Most Reactive Element in the Periodic Table

Elemental fluorine (F₂) is a pale-yellow gas produced by electrolyzing hydrogen fluoride. It is one of the strongest oxidizing agents known.
Fluorine enables the creation of:
- Hexafluorophosphate (PF₆⁻) – essential for lithium-ion battery electrolytes.
- Sulfur hexafluoride (SF₆) – used as an electrical insulator (although declining due to environmental concerns).
- Uranium hexafluoride (UF₆) – used in nuclear fuel enrichment.
- Hydrofluorocarbons (HFCs) – refrigerants and cooling agents.
Modern fluorine chemistry would not exist without fluorspar as the starting raw material.
Hexafluoride Compounds and Industrial Applications
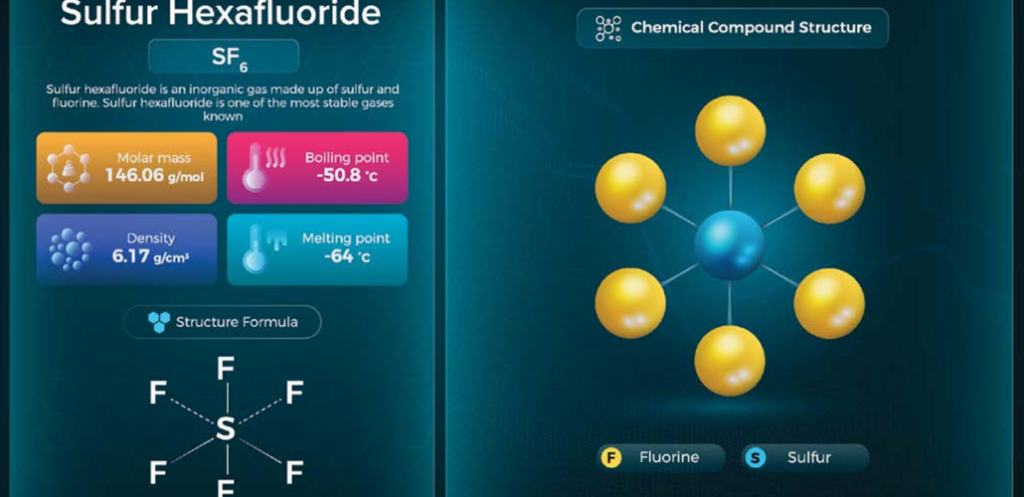

Sulfur Hexafluoride (SF₆)
A non-toxic, non-flammable gas used for:
- High-voltage circuit breakers
- Gas-insulated switchgear (GIS)
- Particle accelerators
However, SF₆ has one of the highest global warming potentials (GWP), prompting global phase-down policies.
Uranium Hexafluoride (UF₆)
Vital to the uranium enrichment process for nuclear fuel.
Hexafluorophosphate (PF₆⁻): The Battery Revolution

Lithium hexafluorophosphate (LiPF₆) is a cornerstone of modern lithium-ion battery electrolytes due to:
- High ionic conductivity
- Stability across broad temperatures
- Good electrochemical performance
Global electrification and EV growth significantly increased demand for PF₆⁻, indirectly driving up long-term fluorspar demand.
Modern fluorine chemistry would not exist without fluorspar as the starting raw material.
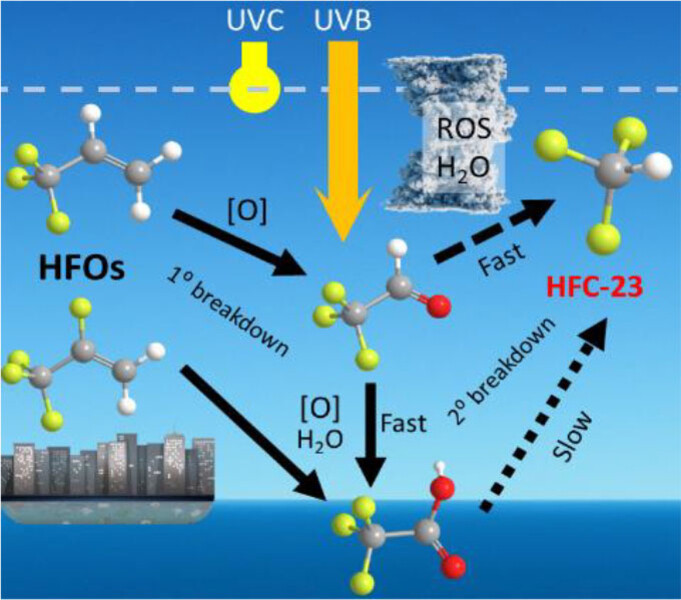
Hydrofluorocarbons (HFCs): Transitioning to Low-GWP Alternatives
Hydrofluorocarbons (HFCs) are widely used in:
- Refrigeration
- Air conditioning
- Aerosol propellants
Common HFCs include R-32, R-125, and R-134a.
Due to climate regulations (Kigali Amendment), industries are shifting toward hydrofluoroolefins (HFOs) and natural refrigerants, but HFC manufacturing still relies heavily on HF produced from acid-grade fluorspar.